This research uses artificial intelligence to predict the progression of Alzheimer’s disease and cancer using medical imaging data. By analyzing brain scans, tumor scans, and treatment responses, AI models can forecast disease development and treatment outcomes, enabling earlier intervention, more personalized care, and improved quality of life for aging populations.
This research develops synthetic genetic circuits that automatically alternate CAR T-cell activity between active cancer killing and recovery states. By preventing immune-cell exhaustion, these circuits could improve cancer immunotherapy effectiveness. The work also suggests broader biomedical applications where controlled cycling of gene activity may enhance treatment safety, longevity, and therapeutic performance.
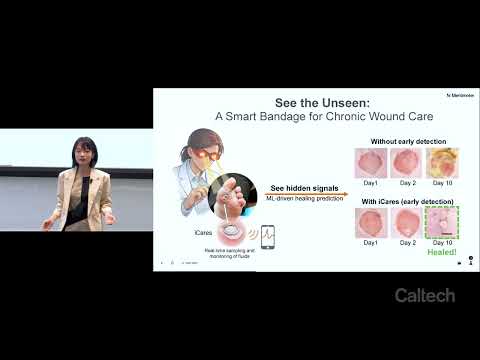
This research introduces iCares, a smart wound-monitoring bandage designed to detect infection and inflammation before visible symptoms appear. Using biosensors, fluid sampling, and machine learning, the system provides real-time wound analysis, enabling earlier intervention, personalized treatment, reduced complications, and improved healing outcomes for patients with chronic wounds.
This research uses spatial transcriptomics to map interactions between T cells, cancer cells, and immunosuppressive cells in tumours. Findings suggest cancer suppresses immune responses by surrounding and weakening T cells. The work aims to improve immunotherapy and enable personalised cancer treatment through detailed tumour mapping.
This research develops targeted radiopharmaceutical therapies for HER2-positive cancers. By attaching radioactive isotopes to trastuzumab, treatment delivers precise radiation to cancer cells, overcoming drug resistance. The work includes creating practical drug kits and aims to improve cancer outcomes by replacing non-specific therapies with highly accurate, targeted interventions.
This talk traces the devastation of the Black Death to highlight a modern crisis: antibiotic resistance. Misuse of antibiotics accelerates the rise of superbugs. Using AI and machine learning, the research identifies genetic resistance patterns and guides effective treatments, aiming to improve clinical decisions and prevent a return to a pre-antibiotic era.
Pagination
- Previous page
- Page 2
- Next page