This research examines stroke risk in sickle cell disease by modelling blood flow in the Circle of Willis. While ultrasound predicts risk in children, it fails in adults. Using MRI-based, patient-specific simulations, the study identifies major differences in blood flow patterns, offering a non-invasive, more reliable method for adult stroke prediction.
Acute respiratory distress syndrome (ARDS) causes severe breathing failure and kills tens of thousands annually, yet has no effective treatment. This research studies how ARDS disrupts lung surfactant, a critical stabilizing substance in the lungs. By identifying immune-related factors that damage surfactant, the work aims to develop the first targeted therapeutic cure.
Tiny errors in electrode placement can determine success or failure of Parkinson’s surgery. This research develops high-resolution Polarization Sensitive Optical Tomography to map brain anatomy at micrometer scale—over 100 times finer than MRI. Automated scanning and 3D reconstruction create detailed connectivity maps, improving surgical precision and neuroscience understanding.
Over 100,000 people await organ transplants, yet preservation limits organs to hours. This research uses radio-frequency sensors to rapidly pre-screen cryoprotective chemicals through dielectric fingerprints, reducing testing from days to minutes. Faster identification of effective preservation agents could extend organ viability and save thousands of lives.
This research presents a new fractional mathematical model for cardiovascular dynamics that maintains the accuracy of traditional methods while greatly reducing complexity. Using only five interpretable parameters instead of twenty, the model analyzes blood pressure in the frequency domain, providing clearer insight into heart function and offering potential improvements for diagnosis and treatment.
This research develops a virtual human model and predictive algorithm to detect blast-induced traumatic brain injuries in real time. Using simulations and body-mounted sensors, the system estimates injury risk on the battlefield, helping medics and commanders make rapid decisions to protect soldiers and improve mission safety.
This research develops an electrochemical sensor to continuously monitor stress by detecting cortisol, a key stress hormone. Using DNA aptamers and nanostructured electrodes, the sensor overcomes traditional detection limits, improving signal strength and durability. The technology offers a noninvasive method for long-term stress tracking to support prevention and treatment.
This research improves the safety of stem cell–derived heart cell therapy for heart failure by engineering a drug-controlled genetic safety switch. The approach prevents dangerous post-transplant arrhythmias while allowing transplanted cells to mature and synchronize with the heart, advancing regenerative alternatives to full heart transplantation.
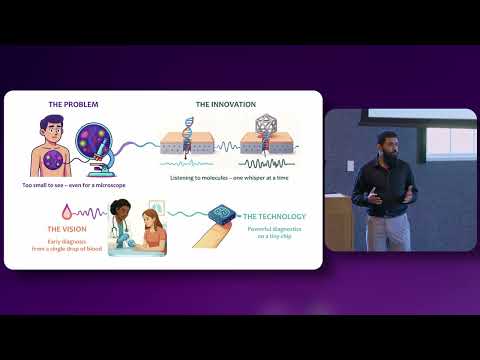
This research develops DNA-origami-enhanced nanopores to detect individual biomolecules from a single drop of blood. By slowing molecules and reading their electrical signatures with machine learning, the technology enables rapid, ultra-early disease diagnosis without traditional laboratory testing.
Healing depends on a balance between extracellular matrix stiffness and cellular recycling through autophagy. This research shows that disrupted balance leads to chronic wounds or fibrotic scarring. By engineering materials with tunable stiffness, the work reveals how cells “sense” their environment, opening new paths to guide healthier wound healing.
Pagination
- Previous page
- Page 2
- Next page