This research develops an electrochemical sensor to continuously monitor stress by detecting cortisol, a key stress hormone. Using DNA aptamers and nanostructured electrodes, the sensor overcomes traditional detection limits, improving signal strength and durability. The technology offers a noninvasive method for long-term stress tracking to support prevention and treatment.
This research develops a nanoparticle-based diagnostic test for thrombotic thrombocytopenic purpura (TTP), a rare and deadly blood disorder. By enabling fast, affordable detection of the ADAMTS13 enzyme, the system could allow earlier diagnosis, timely treatment, and improved survival while inspiring new approaches to rare disease diagnostics.
This research develops a protein-based detection technology capable of identifying subtle molecular changes months before disease symptoms appear. By adapting nanopore sequencing with a protein “detangler,” it enables early warning for conditions like leukemia, shifting medicine from reactive treatment to proactive disease prevention.
Gray mold in strawberries is increasingly resistant to fungicides due to genetic mutations. This research identifies resistance levels by testing pathogen samples in the lab, allowing growers to choose effective treatments. Ongoing work analyzes resistance trends and integrates DNA tools to optimize spray programs and reduce waste, ensuring healthier harvests.
A $2 portable HIV test chip that combines PCR-level sensitivity with home-test simplicity. Using magnetic microparticles, custom probes, and automated processing, it delivers rapid color-change results from a single drop of blood. The system could diagnose HIV and other viruses quickly, affordably, and anywhere.
Prion diseases like CJD are extremely hard to detect early because harmful prions resemble normal brain proteins. This research introduces a new “flashbody” detection tool that binds only disease-causing prions, providing rapid, accurate, equipment-free diagnosis. Early lab results and patient-screening trials are promising, with potential applications to Alzheimer’s and other dementias.
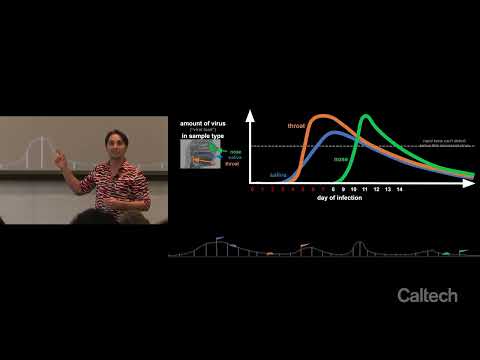
This study tracked viral load in saliva, throat, and nose samples collected daily from newly infected individuals. The findings show each sample type follows a distinct viral-load trajectory, with saliva and throat detecting infection earlier than nose. This has major implications for COVID test accuracy, sampling strategies, and future pandemic preparedness.