This research targets rare genetic diseases caused by frameshift mutations using antisense oligonucleotides as “genetic band-aids.” By masking faulty DNA segments, it restores functional protein production. Demonstrated in muscular dystrophy models, this approach offers a scalable strategy to treat multiple rare diseases, addressing a major gap where most conditions lack effective therapies.
This research applies machine learning to genetic data to distinguish harmless DNA variations from cancer-causing mutations. By treating DNA like a crime scene, the model learns to identify which genetic changes truly drive breast cancer risk, supporting more accurate diagnosis and informed clinical decision-making.
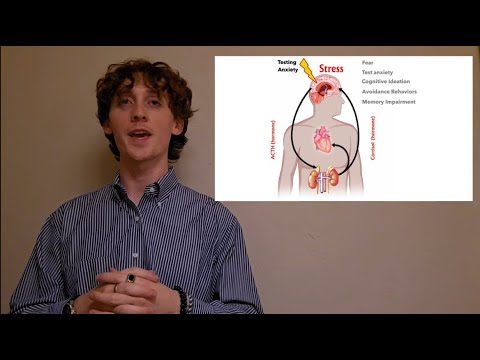
This research explores how chronic stress reshapes the brain through genetic mechanisms. By studying the stress-regulating gene MeCP2 in mice, the work shows how early-life stress can lock the brain into a heightened anxiety state, revealing biological pathways that may inform future treatments for stress-related mental health disorders.
Congenital heart defects are the leading cause of infant death from birth defects. This research develops a high-throughput method to test genetic mutations in key heart genes like TBX5, identifying which variants disrupt heart development. The approach improves diagnosis, informs gene therapy, and advances understanding of why hearts fail before birth.
Craniosynostosis occurs when skull sutures fuse too early, requiring risky surgeries. The researcher identified microRNA-200A as a key regulator of suture development. In mice lacking miR-200A, sutures fused prematurely, but adding extra miR-200A via gene therapy prevented fusion entirely. This breakthrough suggests a non-surgical future treatment for craniosynostosis.
Cleft lip formation may result from broken DNA enhancers—switches that control facial development genes. Scanning the genomes of 130 African children with clefts, this research identified harmful enhancer variants and confirmed their effects in mouse models. The disrupted enhancer likely regulates BMP2, offering new insight into cleft biology and future prevention.
This research focuses on developing reliable blood-based biomarkers to evaluate new treatments for hereditary frontotemporal dementia. By identifying an imbalance between two key molecules, progranulin and prosaposin, the work aims to provide accurate measures of treatment effectiveness and bring hope to families carrying this devastating genetic condition
This research investigates genetic variants that influence how premature babies respond to infections and oxygen shortages—two major causes of brain injury. By analysing the DNA of over 200 premature infants, the study identifies variants linked to later movement and learning difficulties, aiming to enable earlier prediction, prevention, and personalised care.
This research studies the unusually long-lived Heliconius butterflies to uncover genetic mechanisms behind extended lifespan. By analysing DNA from butterflies across their lifespan, it aims to reveal evolutionary strategies for longevity that may inform future human ageing therapies. Understanding diverse animal lifespans could guide healthier ageing — without mythical “Fountains of Youth.”
A hidden evolutionary arms race unfolds between bacteria and the viruses that attack them. By understanding how bacteria cut and rearrange DNA through recombination, researchers can harness these mechanisms for precise gene editing. This work could enable powerful new treatments for genetic diseases, helping patients like the first personalised-therapy recipient, KJ.