This research investigates why blocking an early asthma “alarmin” signal often fails as a treatment. Using mouse models, it reveals that environmental differences—particularly the microbiome—can bypass this signal and still drive asthma. Understanding microbiome health may help predict treatment success and lead to more personalized, effective asthma therapies.
This research investigates Large Granular Lymphocyte Leukemia, where protective T cells become cancerous. The project explores how DNA methylation silences normal T-cell function and tests drugs that reverse this process. By removing harmful chemical modifications, the goal is to restore immune cells to their healthy, protective “superhero” role.
Chickenpox is usually harmless, yet the same virus can cause severe brain infections in some individuals. This research shows that a genetic variant in an immune-system gene reduces antiviral defense, allowing greater viral replication. Identifying such variants helps explain individual vulnerability to severe viral disease.
Type 1 diabetes affects millions worldwide and often begins in childhood, with no cure or prevention. This research uses early-life blood samples and single-cell immune profiling to identify genetic changes in immune cells before disease onset. The findings reveal new biomarkers that could enable early detection, targeted therapies, and future disease prevention.
This research explores how mast cells—immune cells responsible for allergy symptoms—can be repurposed to strengthen vaccines. By targeting mast cells with nasal vaccines, stronger and longer-lasting immune responses may be generated, particularly benefiting high-risk populations and improving protection against infectious diseases.
This research explores how parasitic tapeworms suppress the immune system and how their mechanisms could inspire new treatments for autoimmune diseases. As infections decline, autoimmune conditions rise. Studying rat tapeworm–derived extracellular vesicles, the lab investigates how these molecular signals reprogram inflammatory macrophages, potentially leading to novel therapies that safely regulate immune dysfunction.
This research explores how tissue-resident macrophages guide immature heart muscle cells during early development. By identifying immune signals that enable scar-free heart regeneration in newborns, the work aims to uncover therapeutic pathways that could restore regenerative capacity and improve outcomes for patients with heart disease.
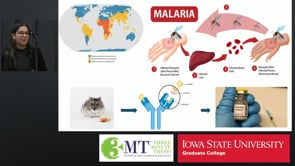
Malaria infects hundreds of millions each year by using the parasite Plasmodium to invade the liver through the CSP protein. This research designs tightly binding antibodies to block infection at its earliest stage, improving vaccine effectiveness and offering a path toward preventing malaria before symptoms begin.
This research examines how macrophages shift between tumor-fighting and tumor-supporting roles in breast cancer. By identifying signals in the tumor microenvironment and engineering molecular cues to promote tumor-destroying behavior, the work aims to reprogram immune responses and improve therapeutic outcomes for breast cancer patients.
Pagination
- Page 1
- Next page