This research challenges the one-size-fits-all approach to obesity by comparing childhood- and adult-onset cases. Through physiological testing before and after weight loss, it examines differences in inflammation, metabolism, and fitness. Findings aim to support personalised treatments, improving patient outcomes and reducing the broader healthcare burden associated with obesity.
This research develops a hybrid drug for dry age-related macular degeneration, combining anti-inflammatory and antioxidant mechanisms. By targeting both inflammation and oxidative stress, it aims to slow disease progression more effectively than existing treatments. Laboratory models test whether the combined therapy outperforms individual or co-administered components in preserving retinal function.
This research explores how immune-related cells and molecules, beneficial in wound healing, may become harmful in Parkinson’s disease. Using the fruit fly as a model organism, the study investigates which inflammatory processes contribute to brain damage. Early results suggest that excessive activation worsens degeneration, offering potential targets for future therapies.
This research investigates why blocking an early asthma “alarmin” signal often fails as a treatment. Using mouse models, it reveals that environmental differences—particularly the microbiome—can bypass this signal and still drive asthma. Understanding microbiome health may help predict treatment success and lead to more personalized, effective asthma therapies.
This talk explores how the modernization of global diets has reduced food diversity and displaced fermented foods, contributing to rising rates of chronic disease. Drawing inspiration from traditional Japanese diets, the research focuses on fermented foods and their impact on gut health and immunity. The speaker highlights the discovery of bioactive, bioavailable cyclic dipeptides in certain Japanese fermented foods, which enhance immune cell function while reducing harmful inflammation. The work suggests that affordable, traditional fermented foods can play a powerful role in supporting immune health and preventing disease.
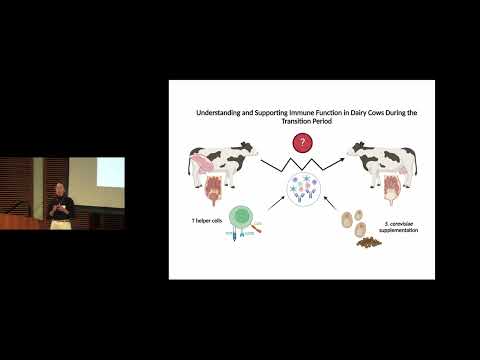
This research examines immune function in dairy cows during the transition from pregnancy to lactation, a period of high disease risk. Focusing on T helper cells, the study shows that yeast supplementation reduces inflammatory activation markers, suggesting a potential strategy to support immune health, improve animal welfare, and reduce disease-related losses in the dairy industry.
Intestinal cells protect us from harmful bacteria by forming a physical barrier and raising immune danger signals when needed. This research reveals a nuclear “knight” molecule that suppresses unnecessary immune activation during metabolic stress, helping maintain intestinal health and preventing excessive inflammation.
This research presents an anti-inflammatory surgical gel that actively reprograms the immune response at the injury site. Rather than masking symptoms, it promotes proper healing, reduces prolonged inflammation, and improves recovery—especially for patients with delayed healing, such as those with diabetes—aligning biomaterials with modern surgical precision.
This research examines how macrophages shift between tumor-fighting and tumor-supporting roles in breast cancer. By identifying signals in the tumor microenvironment and engineering molecular cues to promote tumor-destroying behavior, the work aims to reprogram immune responses and improve therapeutic outcomes for breast cancer patients.
This research shows that doxorubicin disrupts immune signaling between the spleen and heart, priming inflammatory cells that worsen cardiac damage when hypertension develops later in life. Using a two-hit mouse model, the work reveals a heart–spleen axis and identifies immune cells as targets to protect childhood cancer survivors from heart failure.
Pagination
- Page 1
- Next page