This research uses spatial transcriptomics to map interactions between T cells, cancer cells, and immunosuppressive cells in tumours. Findings suggest cancer suppresses immune responses by surrounding and weakening T cells. The work aims to improve immunotherapy and enable personalised cancer treatment through detailed tumour mapping.
This study investigates how immune cells influence Alzheimer’s disease. Using a mouse model, researchers found that removing T cells did not alter amyloid plaque levels but changed microglial behavior, leading to better protection of myelin. The findings suggest T cells may worsen neurodegeneration and highlight new therapeutic possibilities.
This research investigates why blocking an early asthma “alarmin” signal often fails as a treatment. Using mouse models, it reveals that environmental differences—particularly the microbiome—can bypass this signal and still drive asthma. Understanding microbiome health may help predict treatment success and lead to more personalized, effective asthma therapies.
This talk explores how the modernization of global diets has reduced food diversity and displaced fermented foods, contributing to rising rates of chronic disease. Drawing inspiration from traditional Japanese diets, the research focuses on fermented foods and their impact on gut health and immunity. The speaker highlights the discovery of bioactive, bioavailable cyclic dipeptides in certain Japanese fermented foods, which enhance immune cell function while reducing harmful inflammation. The work suggests that affordable, traditional fermented foods can play a powerful role in supporting immune health and preventing disease.
This research uses fruit flies to study the STING immune pathway, revealing how cells detect viral infections. By identifying Nemo as a missing connector protein active only during infection, the work clarifies how immune responses are triggered. These insights may guide future therapies that balance antiviral defense while limiting immune damage.
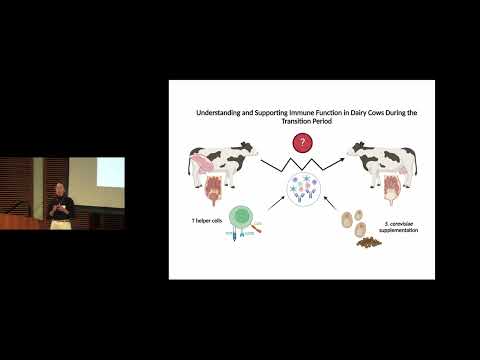
This research examines immune function in dairy cows during the transition from pregnancy to lactation, a period of high disease risk. Focusing on T helper cells, the study shows that yeast supplementation reduces inflammatory activation markers, suggesting a potential strategy to support immune health, improve animal welfare, and reduce disease-related losses in the dairy industry.
This thesis investigates how gut microbes influence brain health through short-chain fatty acids produced from dietary fibre. Measuring these compounds in stool samples, the research finds lower levels in people at risk for Alzheimer’s disease. The next phase tests whether supplementing short-chain fatty acids can prevent or treat Alzheimer’s in mouse models.
Pediatric brain tumors are the leading cause of cancer-related death in children, and current treatments are often insufficient. This research explores harnessing the immune system, particularly macrophages, to fight these tumors. Using advanced imaging and engineered immune cells, the work aims to improve tumor clearance and develop new therapeutic strategies.
This research shows that doxorubicin disrupts immune signaling between the spleen and heart, priming inflammatory cells that worsen cardiac damage when hypertension develops later in life. Using a two-hit mouse model, the work reveals a heart–spleen axis and identifies immune cells as targets to protect childhood cancer survivors from heart failure.
Chronic diseases exhaust the body’s CD8 T cells, weakening their ability to fight infections and cancer. This research identifies CD7 as a key driver of T-cell exhaustion. Removing CD7 keeps T cells active, boosts cytokine production, and improves control of tumors and viruses—offering a promising new immunotherapy target.
Pagination
- Page 1
- Next page