Cancer often becomes resistant to treatment due to the protein CDK8, which helps reprogram cancer cells. Traditional inhibitors fail because CDK8 still acts as a structural scaffold. This research develops targeted degraders that use the cell’s recycling system to eliminate CDK8 entirely, preventing resistance and improving future cancer therapies.
This research uses yeast to study conserved molecular machinery that ensures safe chromosome division. Focusing on a key cohesin-regulating protein, the work reveals how DNA is accurately separated during cell division and how failures in this system can lead to chromosomal errors, developmental disorders, and cancer.
This research uses fruit flies to study the STING immune pathway, revealing how cells detect viral infections. By identifying Nemo as a missing connector protein active only during infection, the work clarifies how immune responses are triggered. These insights may guide future therapies that balance antiviral defense while limiting immune damage.
This research investigates how MRSA loses its antibiotic resistance by shedding the SCCmec genetic element. Environmental stressors such as heat and dryness increase this vulnerability, while antibiotics alone reinforce resistance. Understanding these mechanisms could enable new strategies to reverse resistance and improve treatment options for life-threatening MRSA infections.
This research investigates how cells repair dangerous DNA double-strand breaks through the non-homologous end joining pathway. By identifying key proteins involved in this error-prone repair process, the work reveals new opportunities to sensitise cancer cells to radiation and chemotherapy, potentially improving treatment outcomes for aggressive cancers.
Respiratory Syncytial Virus (RSV) hospitalises thousands of children each year, yet effective treatments remain unavailable. This research investigates a critical protein–protein interaction that enables RSV infection. By identifying and disrupting key molecular binding sites using AI, the work aims to support the development of targeted antiviral therapies for severe RSV.
This research develops a protein-based detection technology capable of identifying subtle molecular changes months before disease symptoms appear. By adapting nanopore sequencing with a protein “detangler,” it enables early warning for conditions like leukemia, shifting medicine from reactive treatment to proactive disease prevention.
Intestinal cells protect us from harmful bacteria by forming a physical barrier and raising immune danger signals when needed. This research reveals a nuclear “knight” molecule that suppresses unnecessary immune activation during metabolic stress, helping maintain intestinal health and preventing excessive inflammation.
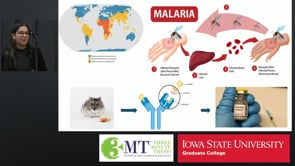
Malaria infects hundreds of millions each year by using the parasite Plasmodium to invade the liver through the CSP protein. This research designs tightly binding antibodies to block infection at its earliest stage, improving vaccine effectiveness and offering a path toward preventing malaria before symptoms begin.
This research develops a rapid, light-based method to study viral fusion, the first step of infection. By applying split NanoLuc technology to HIV, it reveals strain-specific fusion behaviors and unexpected regulatory steps, providing tools that can accelerate responses to future pandemics such as COVID-19.
Pagination
- Previous page
- Page 2
- Next page